The Command Center of Your Distillation Apparatus
I. What is a Distillation Head?
The distillation head is the “brain” and “traffic hub” of the distillation apparatus.
It is a glass three-way joint with a side arm, serving three core functions:
| Function | Description |
|---|---|
| Connection | Connects thermometer above, condenser at the side, and distillation flask below—linking the entire apparatus into a continuous pathway |
| Temperature Measurement | Provides a fixed position for the thermometer to measure the true boiling point of vapor emerging from the distillation flask |
| Flow Direction | Directs vapor smoothly through the side arm into the condenser, where it efficiently condenses into liquid |
The side arm of the distillation head is the “toll booth”—the thermometer bulb must be positioned at the horizontal level of the “toll booth entrance” to measure the true boiling point.
II. Why is the Distillation Head Irreplaceable?
The distillation head is not an optional accessory, but an essential requirement based on scientific principles, experimental safety, operational standards, and data reliability. Its irreplaceability is demonstrated through the following four pillars:
Pillar 1: Achieving Precise Boiling Point Measurement (The Core of Scientific Principle)
This is the most fundamental and irreplaceable function of the distillation head. In organic chemistry, boiling point is a key physical constant for identifying liquid compound purity and identity.
1. Creating a Standard Measurement Point
The junction between the side arm and main chamber of the distillation head is a fixed, reproducible physical location. Literature repeatedly emphasizes that the thermometer bulb must be installed here (aligned with the lower edge of the side arm).
2. Ensuring Measurement of “Equilibrium Vapor Temperature”
At this position, vapor rising from the boiling liquid begins to condense into droplets. The thermometer bulb is completely surrounded by this saturated vapor in gas-liquid equilibrium, giving a reading that represents the true boiling point of the liquid at that pressure.
3. Eliminating Erroneous Readings
- Without a distillation head (e.g., thermometer inserted directly into the flask neck), the thermometer may immerse in superheated liquid (reading too high) or sit in condensate flow (reading too low)—data completely distorted.
- Literature warning: Multiple experimental textbooks explicitly state that incorrect thermometer placement is the most common cause of inaccurate boiling point measurements and separation failures.
Pillar 2: Ensuring Safe and Controllable Experimental Workflow (Security and Efficiency Guarantee)
The distillation head is the structural core of safe, efficient distillation apparatus.
1. Providing Safe, Unobstructed Vapor Pathway
Its side arm is the designated sole pathway for vapor to leave the heat source and enter the condenser. This prevents vapor from escaping through the flask neck or other uncontrollable locations, avoiding burns, fires, or vapor poisoning.
2. Preventing the Apparatus from Becoming a Closed System
The combination of distillation head and thermometer adapter ensures the top of the apparatus remains open to the atmosphere. Multiple experimental textbooks strictly prohibit closed-system distillation, as pressure buildup can cause explosions.
3. Adapting to Complex Operations
- Vacuum Distillation: Must use Claisen distillation head. Its dual-neck design—one for thermometer, one for capillary introducing boiling centers—effectively prevents bumping and flooding under reduced pressure.
- Fractional Distillation: The distillation head sits atop the fractionating column, monitoring and controlling column head temperature and reflux ratio—the command center for precision separation.
- Continuous Operation: By pairing with multi-outlet receivers (such as cow-type adapters), different fractions can be collected without interrupting distillation, improving efficiency and separation.
Pillar 3: Enabling Standardization and Modularity (Foundation of Modern Experimentation)
The distillation head is a key connector in modern standardized ground-glass joint instrument systems.
1. Standardized Joints
Features precise joint sizes (such as 14/20, 19/26, 24/40), enabling quick, tight connections with distillation flasks, condensers, and thermometer adapters of the same standards—eliminating leaks, contamination, and uncertainty from old-style rubber stopper installations.
2. Modular Core
It is the central hub connecting the “vaporization section” (flask), “condensation section” (condenser), and “measurement section” (thermometer). By swapping different “heads” (standard head, Claisen head, fractionating head), you can quickly assemble atmospheric distillation, vacuum distillation, or fractional distillation setups—flexible and reliable.
Pillar 4: Carrying and Transmitting Experimental Technical Standards (Vehicle for Teaching and Research)
The use of distillation heads encapsulates the basic operational standards of organic chemistry experiments.
1. Teaching Priority
How to properly install a distillation head (especially thermometer placement) is a core competency every organic chemistry experimentalist must master and be tested on. Numerous diagrams and exercises in textbooks focus on this.
2. Technical Heritage
From classic textbooks to modern teaching materials, descriptions of distillation head functions and usage remain consistent—demonstrating the heritage and standardization of experimental methodology.
3. Starting Point for Troubleshooting
When distillation shows boiling point fluctuations or poor separation, the first troubleshooting step guided by literature is often: “Check if the distillation head thermometer is installed correctly.”
Summary: Irreplaceability Comparison
| Requirement Aspect | Without Distillation Head | With Standard Distillation Head |
|---|---|---|
| Data Reliability | Inaccurate boiling point measurement, unable to judge product purity | Obtains accurate, reproducible boiling point data |
| Experimental Safety | Apparatus prone to leaks, closure, or flooding—high danger | Provides safe, controllable vapor pathway and open system |
| Operational Efficiency | Tedious, unstable setup; difficult to perform vacuum or fractional distillation | Quick, modular assembly; adapts to various advanced distillation techniques |
| Technical Standards | Random operations; cannot standardize teaching or heritage | Core vehicle for standardized operations, skill training, and troubleshooting |
III. Common Types of Distillation Heads
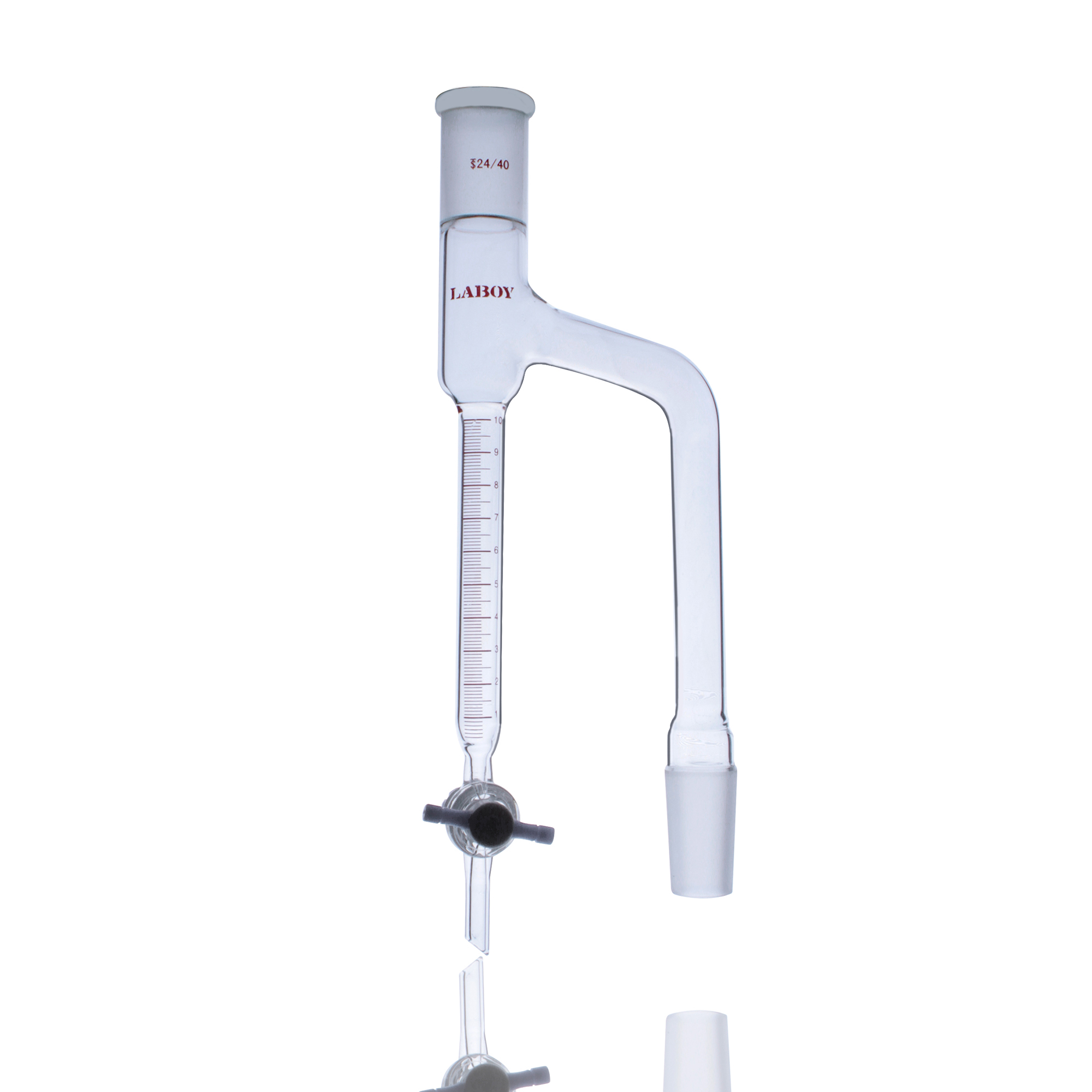
1. Standard Distillation Head
“Y” or “T” shaped design—the first choice for routine distillation
- Features: Single-neck design with side arm and standard thermometer joint
- Applications: Simple distillation, atmospheric distillation, boiling point determination
- Key Specification: The upper edge of thermometer bulb must align horizontally with the lower edge of the side arm
- Literature Sources: Described in “Basic Organic Chemistry Experiments,” “Organic Chemistry Experiments,” “University Chemistry Experiments,” and virtually all experimental textbooks in the “simple distillation” sections

Standard distillation head with condenser—the foundational configuration for routine distillation
2. Claisen Distillation Head
The safety guardian for vacuum distillation
- Features: Dual-neck design—one for thermometer, one for capillary or anti-bumping device
- Core Function: Prevents bumping during vacuum distillation, avoiding liquid flooding into condenser
- Applications: Vacuum distillation of high-boiling point, heat-sensitive compounds
- Literature Sources:
- “Basic Chemistry Experiments 2: Organic Chemistry Experiments”: Detailed description in Experiment 2-9 “Vacuum Distillation of Ethyl Acetoacetate”
- “Organic Chemistry Experiments” (Wang Xueli, Mao Yan eds.): Mentioned in Experiment 8
- “Techniques in Organic Chemistry”: Describes Claisen adapter as distillation head
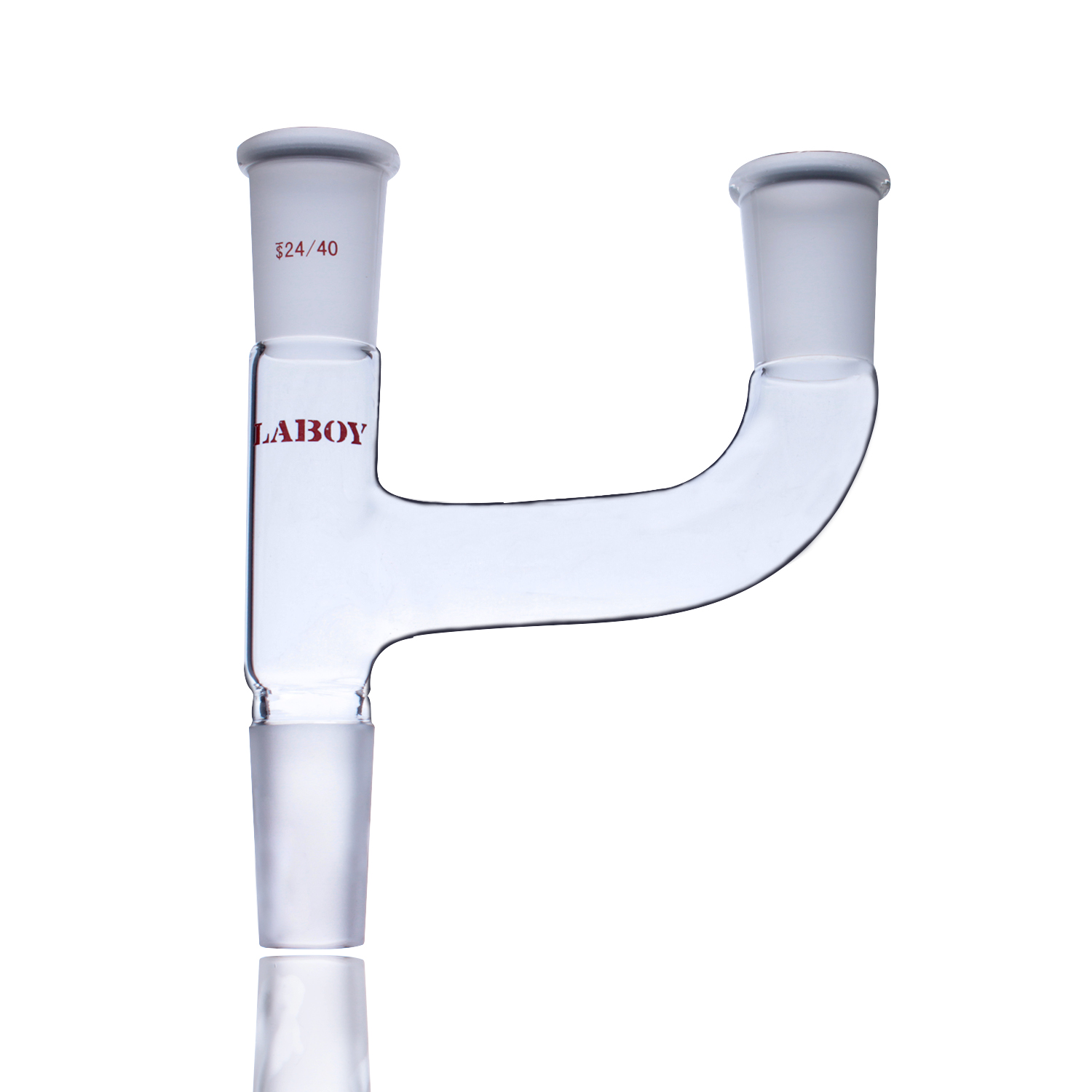
Three-way Claisen adapter—essential safety design for vacuum distillation
3. Short Path Distillation Head
High vacuum, high efficiency precision separation
- Features: Compact design reduces vapor path and pressure drop
- Applications: High vacuum distillation of high-boiling substances, separation of heat-sensitive compounds
- Advantages: Minimizes thermal degradation risk, improves separation efficiency
- Special Types: Vacuum jacketed design for further thermodynamic optimization
Short path distillation head with vacuum jacket—ideal choice for high vacuum operations
4. Microscale Distillation Head
Precision solution for semi-micro experiments
- Features: Compact size, often multifunctional (e.g., doubling as drying tube)
- Applications: Small-scale distillation of precious samples (<5mL), teaching demonstrations
- Advantages: Saves material, quick setup, suitable for high-throughput screening
- Literature Sources:
- “Semi-Micro Organic Chemistry Experiments”: Core content focuses on semi-micro methods
- “Techniques in Organic Chemistry”: Explicitly describes “Williamson microscale Claisen adapter/distilling head,” noting it can serve as drying tube
5. Fractionating Head
Control center for precision separation
- Features: Used in fractional distillation setups; may include condensate traps, fraction collection valves, or multi-outlet receiver interfaces
- Core Functions: Monitor column head temperature, control reflux ratio, direct different fractions
- Applications: Separation of mixtures with close boiling points
- Literature Sources: “Modern Organic Chemistry” (Ichizo Suemura): Implies need for apparatus collecting different boiling range fractions when describing petroleum fractionation
6. Vacuum Distillation Head
Solution for continuous multi-fraction collection
- Features: Multi-outlet design allowing receiver switching while maintaining vacuum
- Core Functions: Vacuum sealing, multi-fraction collection
- Applications: Vacuum distillation (multiple fractions)
- Literature Sources: “Practical Organic Chemistry Handbook” (Li Shuwen, Fan Rulin eds.): Explicitly mentions “Anschütz-Thiele adapter” and describes its function
7. Special Function Distillation Heads
Solvent Still Head
- Applications: Anhydrous solvent distillation, strict water and oxygen removal
- Features: Designed specifically for solvent purification, ensuring oxygen-free environment

Solvent still head—ideal choice for anhydrous solvent distillation
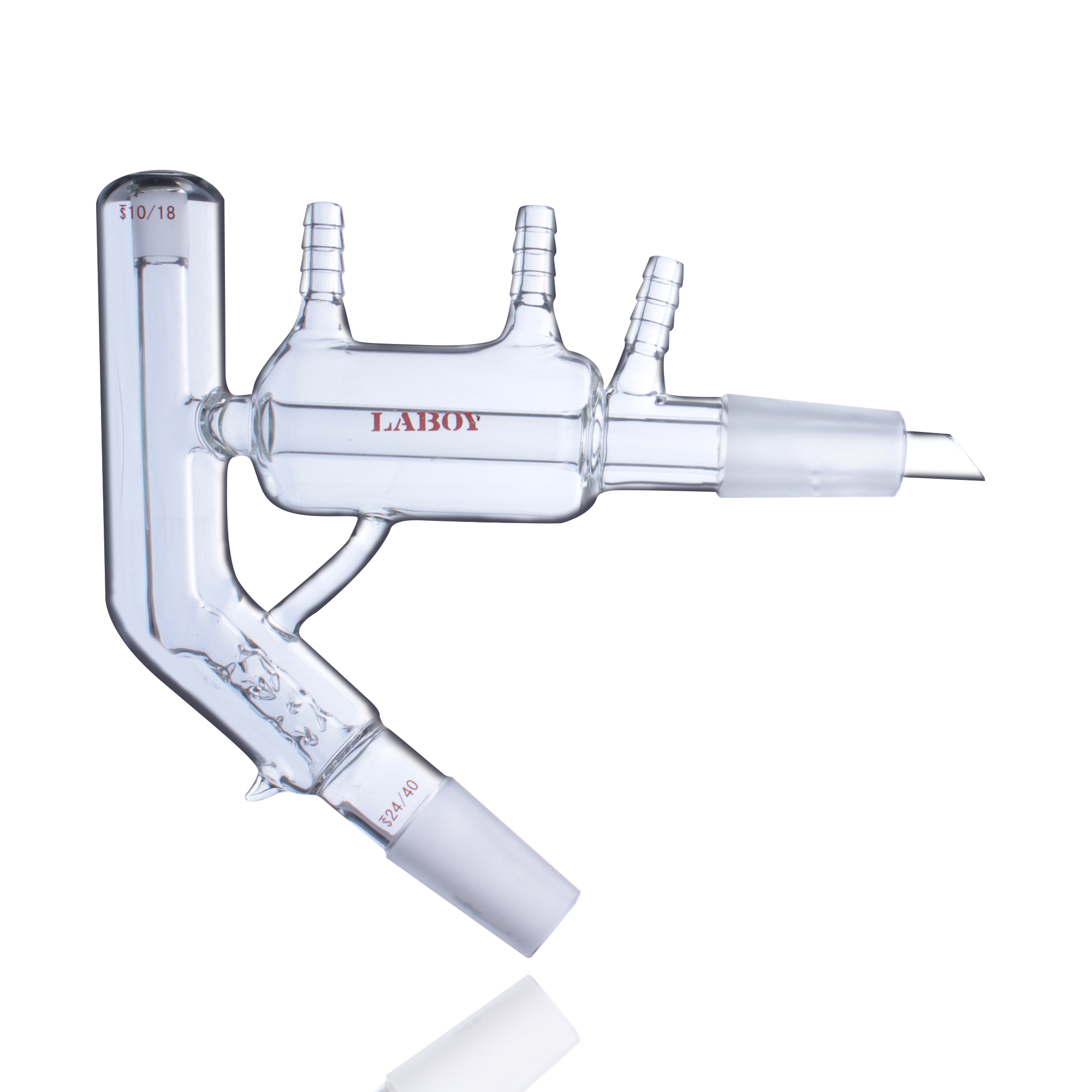
Dean-Stark Apparatus
- Applications: Precise removal of water from reaction systems, water yield determination
- Principle: Uses azeotropic distillation to separate and measure water
Dean-Stark apparatus—standard tool for precise water removal in organic chemistry
Distillation Head with Fraction Collection
- Applications: Continuous multi-fraction collection without interrupting distillation
- Features: Equipped with rotating receivers or multi-outlet collection devices

Three-way cow-type distillation receiver—efficient multi-fraction collection
IV. Complete Workflow Guide for Using Distillation Heads
Phase 1: Pre-Installation Preparation
1. Select Appropriate Distillation Head
- Simple/Atmospheric Distillation → Standard distillation head
- Vacuum Distillation → Must use Claisen distillation head (anti-bumping)
- Semi-Micro Experiments → Microscale distillation head
- High Vacuum Precision Separation → Short path distillation head
2. Inspection and Cleaning
- Confirm glass has no cracks or chips (prevents implosion under vacuum)
- Ensure clean and dry to avoid contamination or bumping
3. Thermometer Preparation
- Range should exceed expected boiling point by 10-20°C
- Temperature calibration recommended
Phase 2: Proper Installation (Critical Steps)
The upper edge of the thermometer bulb must be horizontally aligned with the lower edge of the distillation head side arm.
Why This Matters:
- Correct: Bulb completely surrounded by saturated vapor → True boiling point measured
- Too high: Measures condensate temperature → Reading too low
- Too low: Affected by superheated liquid → Reading too high
Standard Assembly Sequence:
Ring stand → Heat source → Distillation flask → Distillation head → Thermometer → Condenser → ReceiverSpecial Installation for Claisen Distillation Head:
- Main neck connects to distillation flask
- Side neck inserts capillary (adjusted to continuous fine bubbles as boiling center to prevent bumping)
- Thermometer installed in main neck
Phase 3: Operational Essentials
Before Heating:
- Confirm apparatus is open to atmosphere (Never distill in a closed system!)
- Add boiling chips or capillary (Never add after boiling begins!)
During Distillation:
- Control rate: 1-2 drops per second
- Record initial boiling point (first drop) and final boiling point—calculate boiling range (narrower range = higher purity)
Special Procedures for Vacuum Distillation:
- Evacuate first; wait for stable pressure before heating
- At end: Remove heat first, cool system, then slowly admit air to release vacuum
- Never use Erlenmeyer flasks or flat-bottom flasks as receivers (implosion risk!)
Phase 4: Shutdown and Maintenance
Stopping Distillation:
- Never distill to dryness! Stop when small liquid remains or temperature changes significantly
- Remove heat first, then turn off cooling water
Disassembly and Cleaning:
- Reverse order of assembly
- Clean side arm and inner chamber promptly to prevent residue solidification
- Store properly to avoid joint damage
V. Common Errors and Safety Warnings
| Error | Consequence | Prevention |
|---|---|---|
| Incorrect thermometer position | Inaccurate boiling point, separation failure | Strictly align with lower edge of side arm |
| Closed system | Pressure buildup, possible explosion | Ensure open to atmosphere |
| Heating too fast | Bumping, flooding, wide boiling range | Heat slowly, control 1-2 drops/second |
| Wrong receiver (vacuum) | Flat-bottom flask implosion | Use round-bottom flasks |
| Open flame for low boiling point | Fire or explosion | Use water bath/oil bath |
| Forgot boiling chips / adding during boiling | Violent bumping | Add beforehand; never add during boiling |
VI. Quick Selection Guide
| Experiment Type | Recommended Distillation Head | Key Accessories |
|---|---|---|
| Simple Atmospheric Distillation | Standard distillation head | Thermometer adapter |
| Vacuum Distillation | Claisen distillation head | Capillary, vacuum trap |
| Short Path High Vacuum | Short path distillation head | Vacuum jacket, cold trap |
| Fractional Distillation | Head with Vigreux column | Fractionating column |
| Microscale Experiments | Microscale distillation head | Micro flasks |
| Anhydrous Solvent Distillation | Solvent still head | Schlenk flasks |
| Water Determination/Removal | Dean-Stark apparatus | Receiver tube, condenser |
| Multi-fraction Collection | Cow-type receiver set | Rotating receiver, vacuum adapter |
VII. Technical Specifications
Quality distillation heads should feature:
- Borosilicate glass construction: Excellent thermal shock resistance and chemical stability
- Standard joint sizes: 14/20, 19/26, 24/40, etc., ensuring equipment compatibility
- Precision ground joints: Ensuring vacuum tightness and connection reliability
- Hand-blown quality: Ensuring uniform wall thickness, no stress concentration points
VIII. Further Reading and Resources
Recommended Technical Articles:
- “Laboratory Condenser Selection: Don’t Let It Become the ‘Bottleneck’ of Your Experiment”
- “The Invisible Hardware Killers: 5 Reasons Your Synthesis Failed Before It Began”
- “The Physics of Glassware: Why Shape and Joints Matter in Chemistry”
- “The Ultimate Guide to Ground Glass Joints: Sealing, Maintenance, and Emergency Rescue”
- “Your First Organic Chemistry Lab: A Practical Safety Briefing”
- “How to Use Heat Safely in an Organic Chemistry Lab”
Conclusion
The distillation head is one of the most basic yet critical components in organic chemistry experiments. Choosing a well-designed, expertly crafted distillation head not only ensures reliability of experimental data but also safeguards the experimenter.
In the world of organic chemistry, the distillation head is the key physical interface that transforms theoretical principles into reliable experimental techniques. It ensures that from classic experiments a century ago to today’s research, the fundamental operation of “distillation” maintains its scientific validity, safety, and reproducibility.