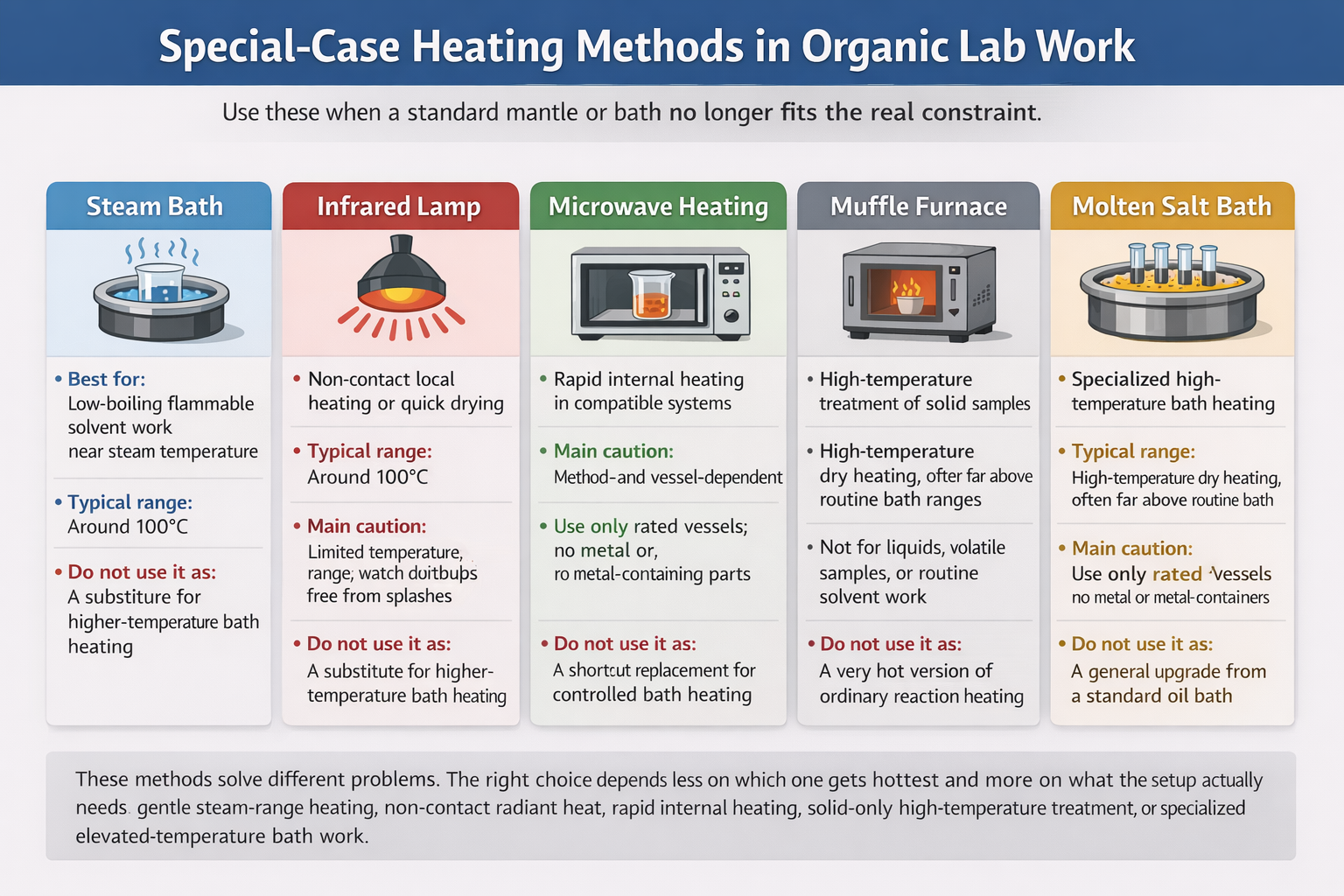
Most organic lab heating can be handled with the ordinary trio: a heating mantle, a suitable heating bath, or a well-matched hot plate setup. Special-case heating methods come in when those standard choices stop fitting the real constraint.
Sometimes the problem is temperature range. Sometimes it is the need to avoid direct flame, avoid bath contamination, keep the heat source off the apparatus, heat a solid at very high temperature, or accelerate a process that responds poorly to ordinary surface heating. These methods are not “better heaters” in a general sense. They are narrower answers to narrower problems.
Start with the real constraint, not the most exotic tool
This topic gets messy when every method is treated as just another box on a temperature chart. At the bench, that is rarely how the decision actually works. What matters first is the job you are trying to do.
Need gentle heating near 100 °C?
A steam bath is often the cleaner answer, especially when the system is flammable and you do not need temperatures above the steam range.
Need rapid internal heating in a suitable reaction medium?
Microwave heating can be extremely effective, but only in equipment and vessels meant for that job.
Need non-contact local heating or quick drying?
An infrared lamp can be useful when line-of-sight radiant heat is what you actually want.
Need much higher temperatures than a routine bath can reach?
Molten salt baths, metal or alloy baths, or a muffle furnace may enter the picture, but each comes with much tighter boundaries than ordinary reaction heating.
That last point matters more than it may seem. A molten salt bath is not just “a hotter oil bath.” A muffle furnace is not just “a very hot oven.” A microwave reactor is not just “faster reflux.” Once you move into these methods, the apparatus logic changes with the heat source.
Steam baths: simple, limited, and often exactly right
Steam baths are easy to underrate because they are so modest. They top out at about the temperature of steam and boiling water, which makes them useless for many hotter operations. But that limit is also their strength. If the task is warming or evaporating a low-boiling flammable solvent, or running a process that should stay near the steam range, a steam bath can be one of the safer and more forgiving choices.
That is why steam baths show up so often in older and still-reliable bench practice for low-boiling solvent work. They do not give you broad temperature flexibility, but they also do not invite you to overshoot in the way an oil bath or aggressively driven mantle can. If your real need is “warm this safely around 100 °C without chasing a precise higher temperature,” the steam bath is doing exactly what it should.
Its limitation is obvious: you cannot ask it to do more than it is built to do. Once the job needs substantially more heat, or tighter control than the steam range can offer, you are already looking for a different method.
Infrared lamps: useful when you actually want radiant heat
Infrared lamp heating is easy to misuse because it looks simple. In reality, it is a line-of-sight radiant heat source with fairly coarse control. That makes it helpful for certain drying tasks, some local warming jobs, and occasional situations where you want the heater off the apparatus rather than wrapped around it or sitting beneath it.
Where it tends to disappoint is temperature uniformity. A bath surrounds. A mantle cradles. An infrared lamp shines. That difference matters. If the vessel geometry is awkward, the receiving surface is uneven, or the lamp distance is guessed rather than judged, heating can be patchy.
It is also not a good method to treat casually. A hot lamp bulb does not tolerate careless splashing, rough handling, or random repositioning once it is already running hot. If you are using an infrared lamp, the correct question is not “can it heat this?” but “is radiant heating actually the right heating behavior for this job?”
Microwave heating: fast when the chemistry and equipment actually fit
Microwave heating is the one method on this page that most clearly changes the pace of the work. In the right system, it can heat very quickly because the energy couples directly into an absorbing medium rather than creeping inward from an external hot surface. That is why microwave-assisted synthesis, digestion, extraction, and fast dissolution workflows can look dramatically different from ordinary bath or mantle heating.
But microwave heating is also the easiest place to blur together three very different things: a domestic microwave, a general lab microwave, and a purpose-built chemistry reactor. For synthetic work, that distinction matters. A dedicated reactor with pressure and temperature control is a different tool from a kitchen microwave with a reaction flask improvised inside it.
The vessel question matters just as much. Metal is a clear no-go in ordinary microwave heating because it can arc. “Microwave-safe glass” also needs to be interpreted properly. Many common borosilicate laboratory glasses are specified under standards such as ISO 3585 or ASTM E438 Type I, Class A, which is useful for understanding the glass material itself. But that does not mean any borosilicate flask is automatically suitable for sealed or pressurized microwave chemistry. Pressure rating, vessel design, cap system, and the reactor’s own operating limits still decide that question.
Microwave heating also has a practical boundary that beginners often miss: some media couple well to microwaves, and some do not. Strongly polar or otherwise microwave-absorbing systems are much easier to heat this way than purely non-polar ones. So if a microwave method looks strangely unresponsive, the problem may not be the power setting. It may be the coupling between the medium and the microwave field.
Muffle furnaces: for high-temperature solid treatment, not ordinary reaction heating
A muffle furnace belongs to a different world from mantles, baths, or lamps. It is an enclosed high-temperature chamber for solid materials that need ashing, calcination, burn-off, or other dry high-temperature treatment. In other words, it is usually for what must happen to the solid itself, not for keeping a flask or solution at reaction temperature.
This is why muffle furnaces show up in ash determinations, burn-off of organics, ceramic or materials work, and certain kinds of residue treatment. They can reach temperatures far beyond routine oil-bath territory, but that is exactly why they are a poor fit for liquid reaction mixtures, volatile solvent work, or anything that still behaves like normal bench-top wet chemistry.
If a sample is still solvent-wet, gives off large amounts of volatile material, or can attack the furnace chamber or elements, a muffle furnace is usually the wrong place to solve the problem. The right move is often to dry, concentrate, or isolate the sample first, then use the furnace only once the sample actually matches furnace logic.
Molten salt baths and metal or alloy baths: high-temperature tools with narrow use windows
Molten salt baths exist for situations where ordinary oil-bath logic runs out of range. They can deliver high, stable bath temperatures that are well beyond the normal comfort zone of routine teaching-lab heating. That makes them useful in specialized high-temperature work, but it also makes them unforgiving. If water, solvent, or incompatible material gets into a very hot molten bath, the result can be violent splattering or worse. These are not casual upgrades for “when you want more heat.”
Metal or alloy baths sit in a similar special-case category. Their appeal is rapid and even conductive heat transfer at elevated temperature. Their downside is that they bring their own handling burden: oxidation, contamination, wetting issues with some materials, and obvious burn risk. In routine organic teaching work, they are much less common than mantles, standard baths, or blocks for good reason.
Comparison table: what each method is actually good for
| Method | Best used when | Main strength | What it is not good for |
|---|---|---|---|
| Steam bath | You want gentle heating near 100 °C, especially for low-boiling flammable solvent work | Simple, limited, relatively forgiving near ~100 °C | Anything that needs substantially more heat or broad temperature flexibility |
| Infrared lamp | You need non-contact local warming or quick surface drying | No bath medium, useful line-of-sight radiant heating | Uniform heating of awkward vessels or precise thermal control |
| Microwave heating | You have a microwave-responsive system and the right reactor-vessel combination | Very rapid internal heating in the right media | Improvised glassware choices, metal parts, or assuming every solvent will respond the same way |
| Muffle furnace | You are treating a solid at high temperature: ashing, burn-off, calcination, or related dry processing | High-temperature enclosed chamber for solids | Ordinary liquid heating, solvent-containing samples, routine reflux, or distillation logic |
| Molten salt bath | You truly need high bath temperatures beyond ordinary organic-lab bath practice | Very high-temperature heat-transfer medium | Routine bench work, beginner operations, or any setup with poor contamination control |
| Metal or alloy bath | You need fast conductive heating in a niche elevated-temperature setup | Rapid, even heat transfer | General-purpose replacement for common baths or mantles |
Common judgment mistakes
Assuming “special” means “better”
Special-case heaters solve constraints. They are not automatic upgrades over a well-chosen mantle or bath.
Forgetting that the heat source changes the apparatus logic
A steam bath, a microwave reactor, and a muffle furnace do not merely change temperature. They change vessel choice, hazard pattern, and what “normal operation” even looks like.
Using a very hot method to solve a modest problem
If a routine bath or better insulation would solve the setup, jumping to molten salt, radiant heating, or a furnace usually means the decision is being driven by drama rather than by bench logic.
Confusing material standards with application approval
A borosilicate vessel may meet a respected glass standard and still be wrong for sealed microwave chemistry or for a harsh thermal cycle in an enclosed furnace.
Where these methods connect to the rest of the setup
Special-case heating choices rarely stand alone. They connect back to vessel shape, condenser behavior, support logic, and what the apparatus is supposed to do with vapor, solids, or pressure once heat is applied. That is why it helps to keep the ordinary setup pages in view while making this decision.
If the real problem is still basic heater choice for a round-bottom flask, start with Heating Mantles or Heating Baths. If the question is broader than a single device, the parent Mixing & Reaction Setup page is usually the better route. If you are still building first-stage judgment around heating, solvent loss, and setup behavior, Starting Organic Lab Work is the better place to begin than any special-case method. And if the apparatus is already behaving badly, go straight to Troubleshooting Organic Lab Work instead of changing heat sources at random.
FAQ
When should I choose a steam bath instead of a mantle or an oil bath?
Choose a steam bath when the real need is gentle heating near 100 °C, especially for low-boiling flammable solvent work. If the setup needs a much higher temperature, faster ramping, or broader temperature control, a mantle or another bath is the more natural choice.
Can I use metal in laboratory microwave heating?
In ordinary laboratory microwave practice, no. Metal or metal-containing parts can arc and create a real hazard. Microwave work should use vessels and accessories intended for that equipment and that method.
Does ISO 3585 or ASTM E438 borosilicate glass automatically make a vessel safe for microwave chemistry?
No. Those standards describe the borosilicate glass material, which is useful and important, but they do not by themselves approve a vessel for sealed microwave reaction conditions. For microwave chemistry, vessel design and pressure-temperature rating still control the decision.
Is an infrared lamp a good replacement for a standard heating bath?
Usually not. An infrared lamp is most useful when you specifically want non-contact radiant heating or quick local drying. It is not the default choice for uniform, controlled heating of a reaction vessel.
Can I put a liquid sample or a solvent-wet sample into a muffle furnace?
That is usually the wrong move. Muffle furnaces are for high-temperature treatment of appropriate solid samples, not for routine liquid heating or solvent-rich materials. If the sample is still wet with solvent, deal with that first before thinking about furnace treatment.