A Note from the Workbench
In most organic labs, a condenser is treated as a generic utility—something you grab from a drawer, hook up to a faucet, and forget about as long as it doesn’t leak. But after 20 years at the glassblowing lathe, I tell my clients this: The condenser is the “traffic controller” of your reaction.
You might smile—it’s just a glass tube, right? Wrong.
If you pick the wrong model or if your cooling capacity can’t keep up with the vapor pressure, you aren’t just losing solvent. You’re creating a “traffic jam” where uncondensed vapor chokes the exhaust path. This leads to a dangerous illusion: your flask is boiling away, but the pressure is silently spiking because the gas has nowhere to go. Before you know it, a joint pops, or worse.
Below is a cheat sheet summarizing my two decades of experience. Scan it first, then let’s dive into the “why” behind these designs.
The “Traffic Controller” Story: A Lesson I Learned 20 Years Ago
I remember a summer afternoon about ten years ago. A professor rushed into my workshop, looking frantic. “Zoe, there’s something wrong with the apparatus you built. The gas won’t escape, the pressure gauge is red-lining, and I’m afraid to keep heating it.”
I went to his lab. It was a scorching day, and the room smelled strongly of diethyl ether. He was using a 500ml flask with a long Liebig condenser. The flask was boiling violently, but no liquid was dripping back.
I touched the water jacket—it was warm. He was using the building’s tap water, which had been sitting in pipes under the sun all morning; it was at least 28°C.
“Professor,” I said, “the tube isn’t blocked. It’s congested.” Diethyl ether boils at 34.6°C. Trying to condense it with 28°C water is like trying to stop a crowd with a suggestion. The uncondensed vapor was filling the entire tube like a wall of people standing in the middle of a highway. The byproduct gases couldn’t get past the solvent vapor.
We switched him to a chilled water circulator. Within two minutes, a neat line of droplets appeared, the exhaust started “burping” bubbles normally, and the pressure dropped instantly. He thought “water is water.” But in reality, if the cooling doesn’t match the chemistry, the condenser is no longer an assistant—it’s a bottleneck.
Six Condensers, Six Personalities
To a glassblower, every condenser has a “temperament.” Pick the right one, and your experiment purrs; pick the wrong one, and you’re asking for trouble.

1. Liebig: The Reliable “Straight-Shooter”
The Liebig is the baseline. A straight inner tube inside a water jacket. It’s a “straight-through” highway for vapor.
- The Design: It’s built for distillation. Because the tube is straight, there are no “pockets” for liquid to get trapped in as it flows toward your receiving flask.
- Pro Tip: It’s cheap and the easiest to clean. But I’ve seen people try to use it for refluxing DCM or ether. Don’t. The surface area is too small; if the vapor flow is even slightly fast, the Liebig simply can’t catch it.
2. West: The “Slim-Fit” Speedster
The West looks like a Liebig that went on a diet. The water jacket is extremely narrow, sitting right against the inner tube.
- Why the “Slim” Design? The narrow jacket forces the cooling water to move at high velocity. Faster water means more aggressive heat exchange.
- Best Use: This is your best friend for vacuum distillation. When you need to maintain a sharp temperature gradient, the West reacts much faster than a bulky Liebig.
3. Allihn: The Vapor “Speed Bump”
You’ll recognize the Allihn by that string of glass “bulbs” inside. These bulbs act as speed bumps: vapor hits the bulb, the space expands, the velocity drops, and the vapor has more time to hug the cold glass walls.
- The Golden Rule: Never tilt an Allihn for distillation. Those bulbs are shaped like cups. If you tilt it, the condensed liquid pools in the bottom of the bulbs instead of flowing out. This chokes the vapor path and causes pressure spikes.
- Best Use: Standard reflux. If you’re heating a reaction overnight at 80°C, the Allihn is your workhorse.
4. Graham: The “Labyrinth” (Handle with Care)
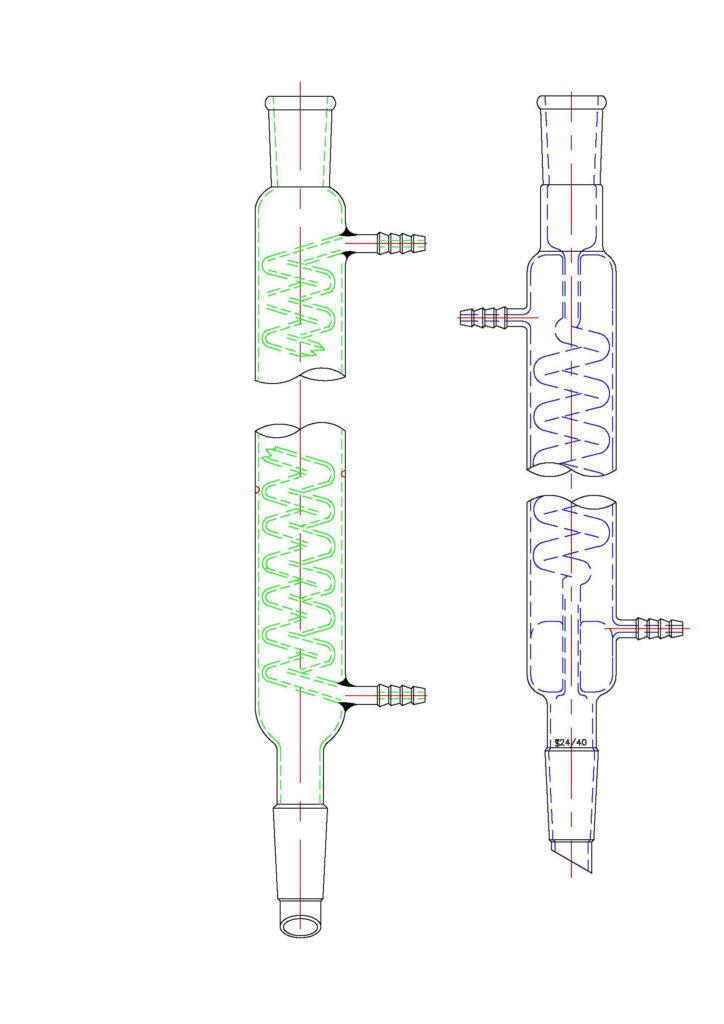
The Graham features a long, coiled inner tube. The vapor has to travel a marathon distance to get through.
- The Warning: ABSOLUTELY PROHIBITED for vigorous reflux. * The Why: That coil is a narrow, winding road. If you have high vapor volume, the liquid trying to drain down will collide with the vapor trying to push up. It’s a recipe for a “puke” (flooding). I once saw a Graham tube pop like a champagne bottle because someone tried to reflux toluene in it. The Graham is for slow solvent recovery only.
5. Dimroth: The King of Reflux

People often confuse this with the Graham, but it’s the opposite: the water goes inside the coil, and the vapor stays in the big, open outer jacket.
- Why it Wins: The vapor has plenty of room to move without resistance, while the cold coil provides massive surface area. It’s the “V12 engine” of condensers.
- Pro Tip: Go for the version where both ports are at the top. This ensures the coil is always 100% full of water with no air pockets, making it the separation efficiency champion for low-boiling solvents like ether.
6. Cold Finger: The “Scalpel”
Just a simple “finger” of coolant dipped into the system.
- The Edge: It’s a space-saver. If you’re doing microscale chemistry or a sublimation, this is it. The product “grows” right on the finger, and you just pull it out and scrape it off when done.
The Quick Decision Matrix
I recommend printing this out and taping it to your fume hood.
| Condenser Type | Structure | Best Used For | Glassblower’s Advice |
|---|---|---|---|
| Liebig | Straight tube jacket | Simple distillation | Cheap and reliable, but inefficient for reflux. |
| West | Narrow jacket, high-velocity flow | Vacuum distillation | Best for high-vacuum stability. Fastest heat exchange. |
| Allihn | Bulb/Bubble structure | Standard reflux | Do not tilt! Vertical use only to avoid liquid trapping. |
| Graham | Inner coil (vapor path) | Slow recovery | Safety Risk: Never use for vigorous reflux due to pressure buildup. |
| Dimroth | Inner coil (coolant path) | High-efficiency reflux | The King. Perfect for low-boiling solvents like ether. |
| Cold Finger | Compact probe | Sublimation / Microscale | Unmatched space efficiency. Great for product collection. |
When refluxing high-volatility solvents like Diethyl Ether or DCM (Dichloromethane), standard Liebig or Allihn condensers often fail to prevent solvent loss. The professional choice is the Dimroth Condenser. Its internal cooling coil provides a massive surface area in a compact space, effectively trapping vapors that would otherwise escape. For maximum efficiency, ensure your coolant temperature is at least 20°C below the solvent’s boiling point to avoid “vapor displacement” issues.
Condenser flooding (or “puking”) usually happens when vapor volume exceeds the condenser’s capacity or when using an incorrect model. If you use a Graham (Coiled) Condenser for vigorous reflux, the narrow spiral path creates a “traffic jam” between the rising vapor and the descending condensate. To fix this, reduce the heat immediately and switch to a wider-bore condenser like an Allihn or Dimroth, which allows for better counter-current flow without pressure buildup.
If you notice solvent vapors reaching your pump during vacuum distillation, your heat exchange rate is likely too low. The West Condenser is specifically engineered for this. Its narrow water jacket forces coolant to flow at a higher velocity, significantly increasing the cooling power compared to a standard Liebig. This ensures vapors are fully liquefied before they can reach the vacuum port, protecting your pump and improving recovery rates.
This is a fundamental rule of laboratory safety and physics. Feeding water from the bottom ensures the condenser jacket is completely filled, eliminating air pockets (air locks) that cause “hot spots.” These hot spots can lead to uneven cooling and thermal stress, potentially cracking the glass. By following the counter-current exchange principle, you ensure the coldest water meets the coolest vapor at the top, creating the most efficient temperature gradient.
Condensers are prone to seized joints because the constant cooling can cause joints to contract or trap solvent. To prevent this, always apply a thin layer of high-quality vacuum grease
or use a PTFE sleeve before assembly. If a joint is already stuck, never use force. Gently warm the outer joint with a heat gun to trigger thermal expansion, or use a dedicated joint gripper. For delicate Dimroth coils, avoid mechanical scrubbing; use a solvent soak or an ultrasonic bath to maintain the internal glass integrity.
Three Mistakes That Drive Glassblowers Crazy
- Backward Water Flow: Remember: Bottom-In, Top-Out. This ensures the tube is actually full. If you go top-down, the water just trickles down the side, leaving half your condenser dry and prone to thermal shock.
- The Graham Myth: Don’t assume “more coils = better reflux.” In a Graham, more coils just means a higher chance of a “puke” or an explosion.
- Tilting the Allihn: I’ve seen PhDs do this. If you need to distil, grab a Liebig. Don’t make the Allihn do a job it wasn’t built for.
Final Thoughts: Take Care of Your Glass
A Dimroth is a work of art, but it’s fragile. The coil connections are thin—don’t go shoving a stiff brush down there. Soak it in solvent or use an ultrasonic bath. And for heaven’s sake, use a little grease on those ground glass joints. I hate seeing beautiful condensers smashed because a joint seized up.
A good condenser can last you twenty years. If you treat it right, it’ll keep your solvents—and your safety—exactly where they belong.
Make sense? Now go give your setup a once-over.
